Published on 06 May 2026
Solvent-Mediated Microfluidic Generation of Cell-Sized Liposomes
This Research Summary article is based on the papers titled “Facile and versatile PDMS-glass capillary double emulsion formation device coupled with rapid purification toward microfluidic giant liposome generation,” authored by Bakouei M, Kalantarifard A, Sundara Raju I, Avsievich T, Rannaste L, Kreivi M, Elbuken C., and “Solvent-Mediated Dewetting Principles for Cell-Sized Liposome Formation,” authored by Bakouei, M.†, Avsievich, T.†, Sundara Raju, I., Stumpf, D., Kalantarifard, A., Ryplida, B., & Elbuken, C.
Introduction to Cell-sized Liposome
The reconstitution of cell-sized liposomes as fundamental building blocks for synthetic cells is of critical importance with far-reaching applications in cell biology, biomanufacturing, and therapeutics. Microfluidic technologies enable precise control over the formation and composition of these synthetic cells, offering throughput and monodispersity superior to conventional generation methods. However, microfluidic liposome production is often hindered by fabrication complexity and the intricate dynamics of the generation process. Here, the team demonstrate a hybrid microfluidic double emulsion formation chip and a solvent-mediated dewetting method aimed to streamline liposome generation for application in synthetic biology. Furthermore, this approach explains dewetting-based, cell-sized liposome assembly.
Double emulsion for liposome generation Setup
Materials:
- Microfluidic hybrid formation chip (Produced via microfabrication using 3D printer mold, Form 3+ Formlabs and standard soft lithography)
- DMIL LED inverted microscope (Leica)
- FastCam mini high-speed camera (Photron)
- OB1 MK3+ Pressure Controller (Elveflow)

Hybrid microfluidic double emulsion formation chip
Bakouei et al. developed a PDMS-glass capillary hybrid chip as a treatment-free and versatile approach for generating double emulsions (DEs), offering an alternative to standard PDMS chips and glass-capillary co-flow devices.
This hybrid architecture leverages the inherent surface properties of both PDMS and glass to provide the
- spatial hydrophobicity and
- spatial hydrophilicity
required for DE formation (Fig. 1), thereby eliminating the need for intricate surface treatments that suffer from a lack of repeatability and short longevity.
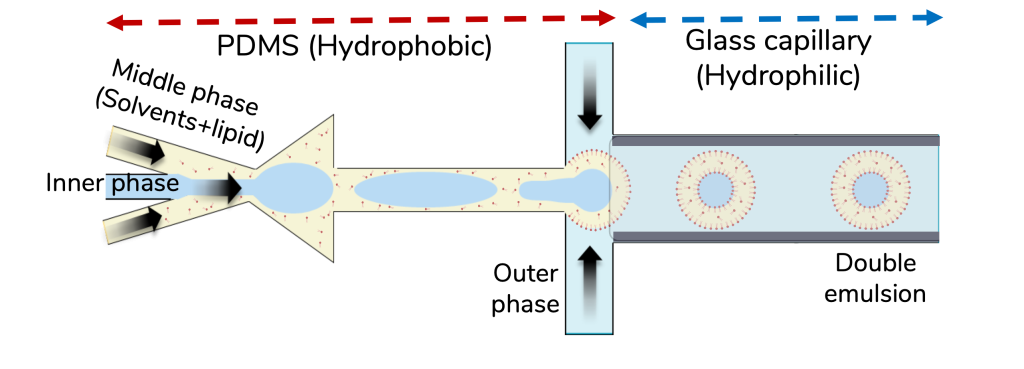
Consequently, the hybrid chip enables DE generation in a simplified, reusable, and versatile manner. Fig. 2a illustrates the two-step assembly of the hybrid chip:
- two identical patterned PDMS replicas are aligned and bonded
- a tapered capillary with specific tip dimensions is inserted into the PDMS housing.
The assembled chip features two consecutive flow-focusing junctions.
- The first junction, situated within the naturally hydrophobic PDMS, facilitates water-in-oil (W/O) droplet formation.
The second junction, located within the hydrophilic glass capillary, facilitates the high-throughput formation of water-in-oil-in-water double emulsions (W/O/W) (Fig. 2b).

Inlet tip diameter influences the size of the double emulsion
As shown in Fig. 3 and Movie 1, by modulating the inserted capillary tip diameter, a wide size range of monodisperse thin-shell double emulsions can be generated, spanning from 1.2 mm down to 27 µm.

Pressure influences the shell thickness
The shell thickness and topology of the DEs can be precisely controlled by modulating the pressures applied across the three phases and by selecting specific formation modes. As demonstrated in Movie 2, the hybrid chip functions under three formation modes.
The double-dripping mode is recommended for the high-yield production of Cell-sized thin-shell DEs.
Key Findings
Solvent-mediated dewetting transition from double emulsion to partially dewetted liposome
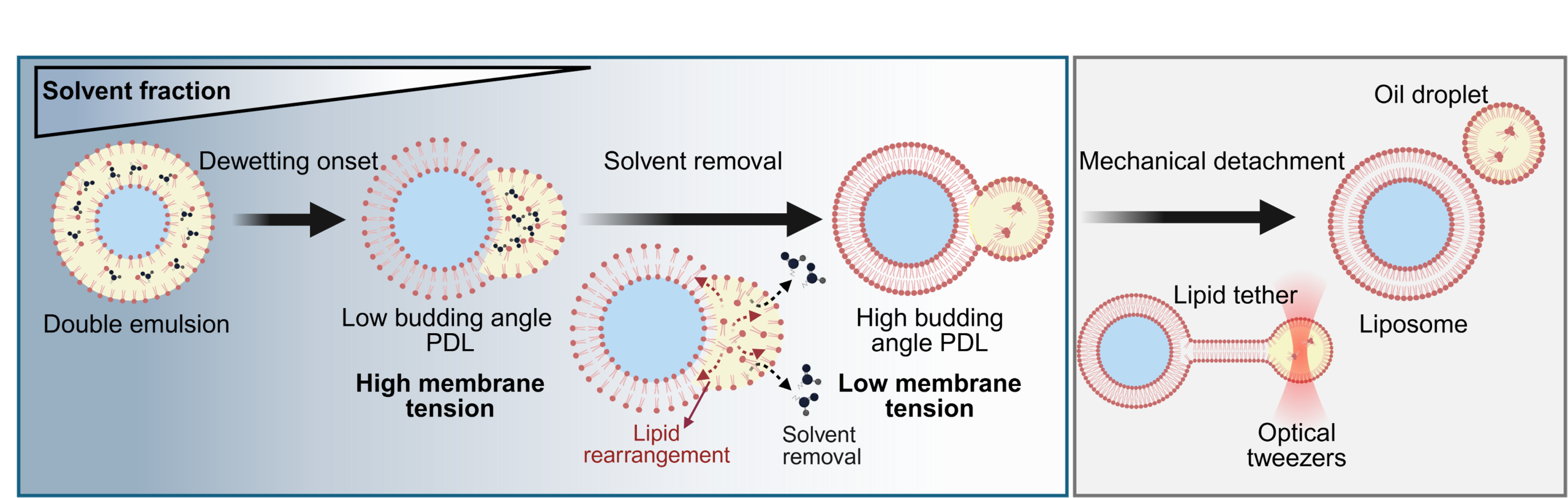
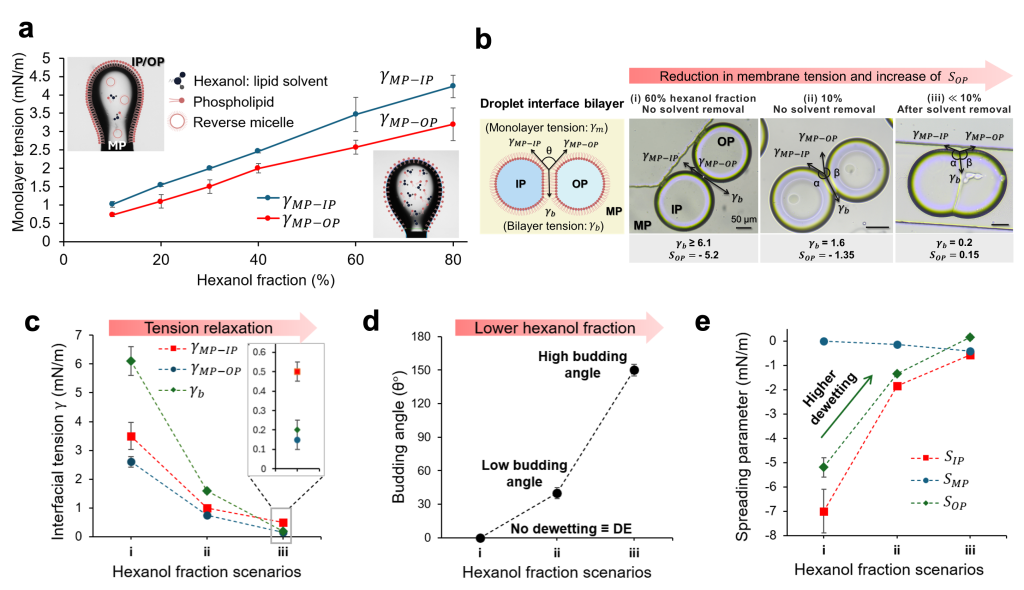
The team elucidates that the dewetting transition from microfluidically generated DEs to liposomes is driven by solvent removal. DEs were generated using a mixture of 60%:40% combination of hexanol and paraffin oil as the solvents of DOPC lipids in the middle phase (MP). Upon formation, the hexanol, being water-soluble and volatile, starts partitioning out of the DE shell. This solvent removal triggers the spontaneous onset of dewetting, resulting in a partially dewetted liposome (PDL) (Movie 3), which is characterized by the contact angle of its residual oil pockets, referred here as the budding angle.
In a semi-permeable glass chamber, solvent removal proceeds extremely slowly, leading to spatial variations in dewetting progression (Fig. 4). DEs situated near air-exposed regions experience a faster dewetting transition, resulting in a high budding angle morphology (q > 90°). In contrast, those lacking air exposure undergo a slower dewetting transition, resulting in a low-budding-angle morphology (q < 90°).
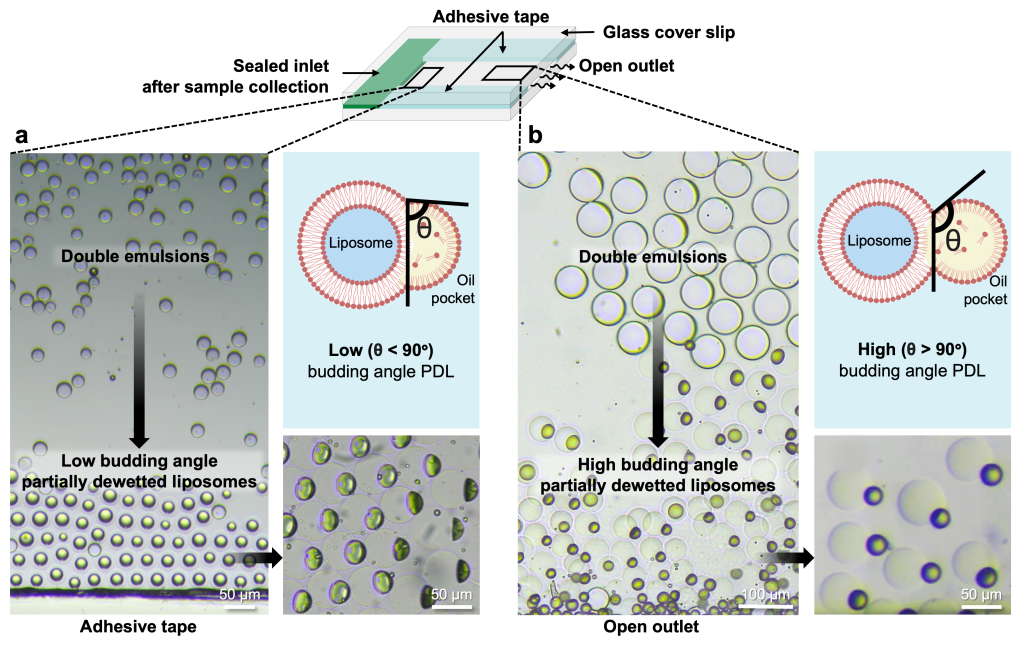
To address the slow and spatially non-uniform dewetting transition, a gas-permeable PDMS chamber was utilized for DE collection. Incorporating a thin PDMS membrane in the chamber ensures a high hexanol removal flux. This approach resulted in a spatially synchronous dewetting transition from DEs to high budding angle PDLs within 10 minutes (Fig. 5 and Movie 4).
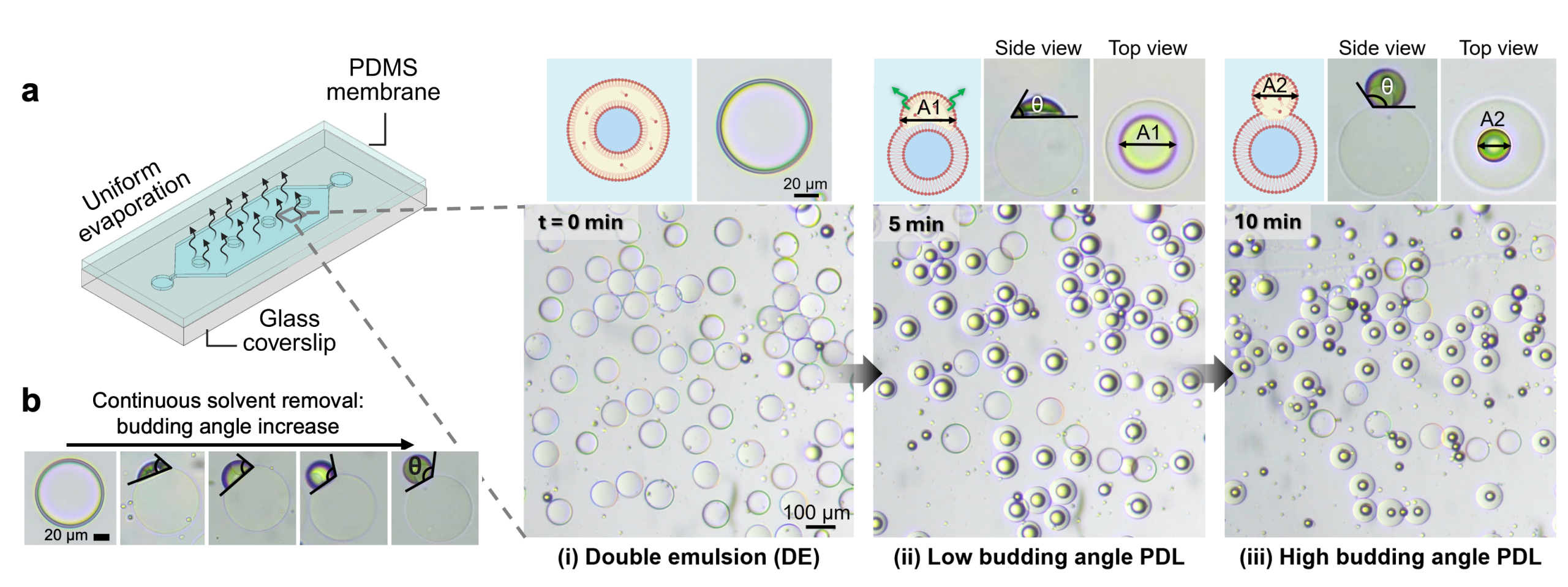
Origin of dewetting morphological transformation upon solvent removal
To elucidate the mechanism underlying the dewetting transition from double emulsions (DEs) to high budding angle partially dewetted liposomes (PDLs) via solvent removal, the process within the framework of ternary phase wetting theory was analyzed.
It was hypothesized that solvent removal simultaneously reduces the interfacial tension of both
- oil-aqueous monolayers (Fig. 6a) and
- the bilayer membrane (Fig. 6b).
This significant reduction in tension, with the membrane tension decreasing more rapidly than the monolayer tensions (Fig. 6c), establishes a new equilibrium, resulting in an altered dewetting morphology (Fig. 6d).
Consistently, the analysis of the spreading parameters, predictive of wetting morphology, demonstrates that solvent removal increases the outer phase spreading parameter ( ), thereby favoring dewetting transition (Fig. 6e).
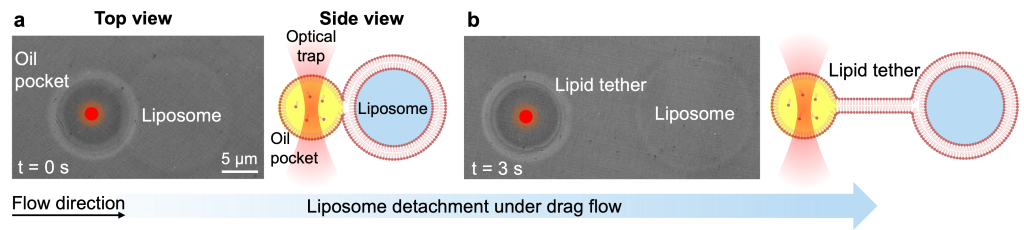
Non-spontaneous nature of complete dewetting
Solvent removal facilitates the morphing of almost fully budded liposomes; however, the liposome and oil pocket maintain a shared neck that keeps them adhered. This suggests that complete detachment is not spontaneous and fission requires an external force.
A characterization of the residual adhesion and the magnitude of the mechanical force required for complete dewetting was then performed. Using optical tweezers, the formation of lipid tethers anchoring the oil pocket was observed, which require a detachment force on the piconewton scale to achieve separation (Fig. 7 and Movie 5).
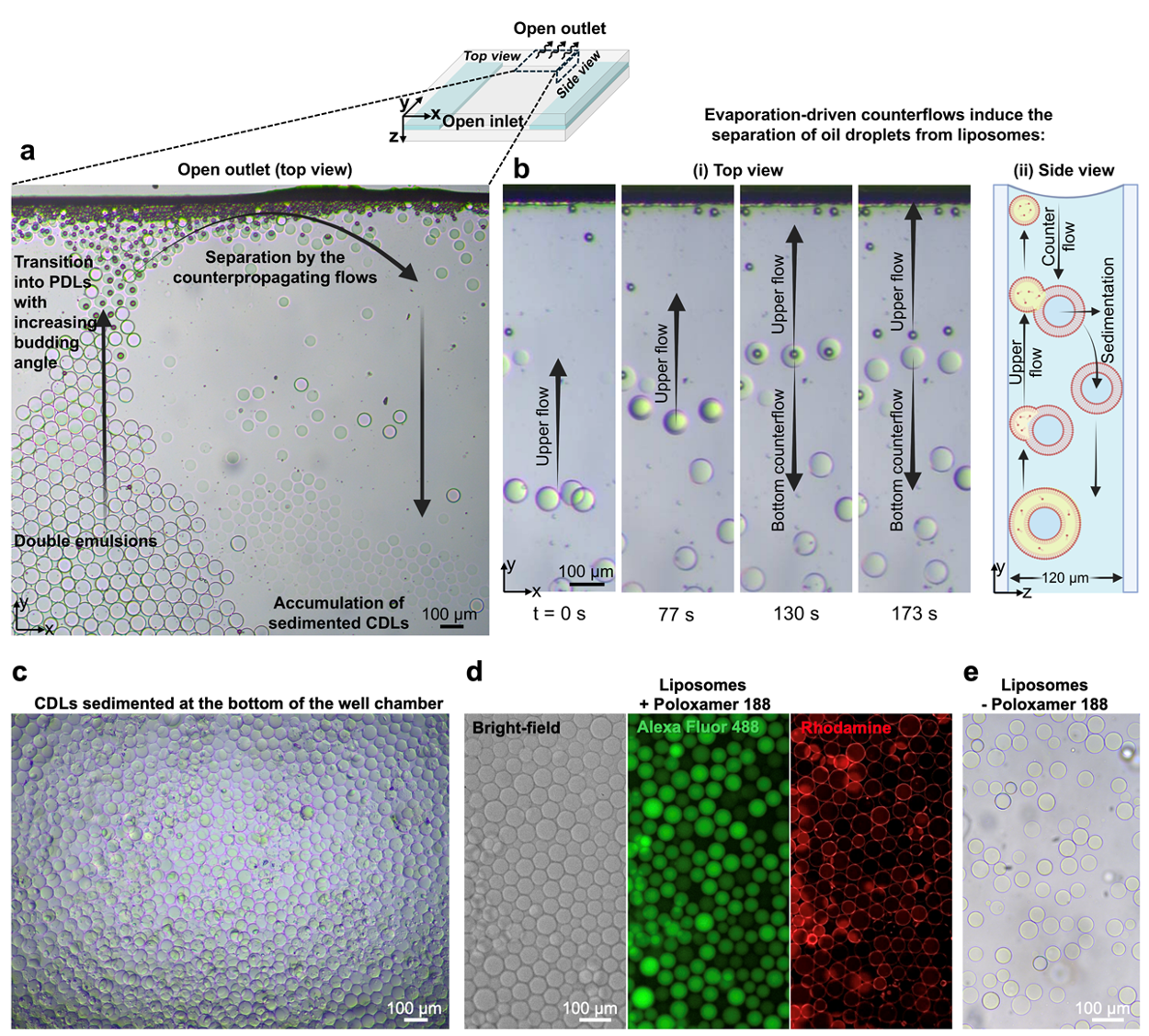
High-yield liposome generation with complete dewetting transition
Solvent removal and mechanical force are essential mechanisms for dewetting and liposome formation. Through evaporation-driven flow and buoyancy-induced separation, high-throughput liposome generation was achieved. Complete separation of the liposomes from the oil pockets was enabled by
- hexanol evaporation and
- shear flows induced by the evaporation of the bulk outer phase (Fig. 8a,b).
While hexanol depletion promoted the dewetting transition to a high budding angle PDL, concurrent evaporation of the outer phase generated internal flows. Sufficient shear forces were thereby created to enable complete dewetting.
Similarly, efficient formation was observed when DEs were collected into a large open-well chamber immediately after generation (Fig. 8c,d). Hexanol evaporation from DEs floating at the surface was facilitated by the lidless chamber. Once a high-budding-angle morphology was reached, pulling stress was applied by buoyancy forces at the oil pocket–membrane interface, promoting complete dewetting. Additionally, liposomes were formed in the absence of surfactants (Fig. 8e), offering greater biocompatibility, as membrane properties may be adversely affected by surfactants, thereby inhibiting biomolecular interaction with synthetic cells.
Conclusion
Here, a hybrid formation chip and solvent-mediated dewetting method are offered as a microfluidic cell-sized liposome generation system.
- Hybrid chip by leveraging PDMS and glass capillary inherent properties enables the treatment-free, versatile, and reusable formation of double emulsion templates.
- The solvent-mediated dewetting enables morphing of liposomes from these templates through solvent removal and mechanical force mechanisms.
The extent of solvent removal governs the oil droplet-liposome dewetting progression and equilibrium morphology. Solvent removal through alteration in monolayers and membrane tension enforces new equilibrium morphologies, favoring dewetting. This research highlights that completion of solvent removal results in an almost budded oil droplet with an attached necking requiring a pN-scale external force for detachment, identified using optical tweezers. This work demonstrates a predictable and high-throughput approach for microfluidic cell-sized liposomes generation in a biocompatible manner.
References
- Bakouei M, Kalantarifard A, Sundara Raju I, Avsievich T, Rannaste L, Kreivi M, Elbuken C. Facile and versatile PDMS-glass capillary double emulsion formation device coupled with rapid purification toward microfluidic giant liposome generation. Microsyst Nanoeng. 2024;10(1):183
- Bakouei, M., Avsievich, T., Sundara Raju, I., Stumpf, D., Kalantarifard, A., Ryplida, B., & Elbuken, C. Solvent‐Mediated Dewetting Principles for Cell‐Sized Liposome Formation. Small, 2026;22(18): e12610.
Additional Information
Authors Bio:
Caglar Elbuken: Professor at the Faculty of Biochemistry and Molecular Medicine, University of Oulu, and VTT technical research center of Finland, and head of the Microfluidics and Biosensor research group
Mostafa Bakouei: PhD researcher at the Faculty of Biochemistry and Molecular Medicine, University of Oulu


Written and reviewed by Mostafa Bakouei, and Louise Fournier, PhD in Chemistry and Biology Interface. For more content about microfluidics, you can have a look here.

 Job
Job Collaborations
Collaborations Customer
Customer Other
Other
