Published on 30 July 2020
Dynamic cell culture
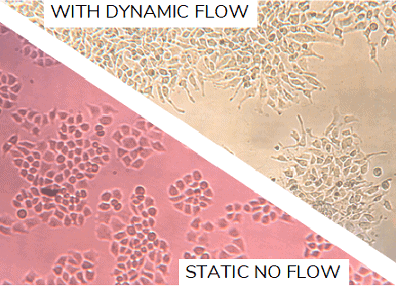
Dynamic cell culture describes the in vitro culture of cells in the presence of applied mechanical stress. Of great emerging interest is the ability to control the flow of fluid, such as medium, over cells while they grow. Flow rate can be generated very precisely using microfluidic instruments and is an ideal way to better replicate the dynamic environment that cells normally reside in. Medium microfluidic perfusion mimics the flow of blood, allowing nutrient exchange and the removal of waste products, and also adds shear stress to cells.
Watch the webinar by our researcher Lisa Muiznieks about dynamic cell culture!
Advantages of this setup for cell biology:
In this application note we describe how to perfuse cells for dynamic cell culture. Perfuse with the automated OB1 pressure controller using a variety of flow profiles, including constant flow or intermittent, steady rate or pulsatile, or create a customised profile, e.g. to mimic the cardiac rhythm. Read also our application notes about automated cell seeding and cell staining for dynamic cell culture.
Some main applications of dynamic cell culture include:
- Live cell imaging (e.g. calcium imaging, FISH)
- Drug screening
- Shear stress
- Cell rolling-adhesion assay
- Immune response
- Cancer invasion and metastasis
- Models of physiology and disease
- Organs on chip
- Blood vessel formation & occlusion (atherosclerosis)
- Bone homeostasis and disease (osteoporosis)
- And many more!
List of components for microfluidic perfusion setup

Microfluidic chamber designed for perfusion
(IBIDI µSlide I Luer)
Microfluidic perfusion setup diagram

Hardware
- Pressure & Flow controller: Impose a given pressure in order to create a stable and pulseless flow.
- Reservoirs: Contain your medium, buffer, stains, cell suspension or samples. Various cap sizes are available, from Eppendorf to bottles.
- Rotation valve (MUX-Distributor): Select the injected liquid.
- Flow Sensor: Monitor and control the flow rate in real time.
- Bubble trap: Trap and remove air bubbles to avoid them entering your chip, disturbing flow and adhered cells.
- Perfusion chamber or microfluidic chip: Contains your cells. Compatible with microscopy.
- Computer: Control all the parameters with our software and automate your experiment by creating injection sequences.
Chemicals
- Cells: HeLa (1×106 cells/mL)
- Medium: DMEM high glucose, 10% FBS, Penicillin/ Streptomycin (100 U/mL; 100 µg/mL)
- Wash buffer: PBS
- Live/ dead cell stains: Calcein/ Hoechst/ Propidium Iodide
[ooc_rebound]
Microfluidic perfusion protocol

Follow our guide to seed and adhere cells in a microfluidic chip.
TIP: To ensure solid attachment, leave cells 12-18h after seeding before starting flow
Perfuse: Switch flow to reservoir containing medium 1.
TIP: use a step gradient to slowly increase flow rate to desired when scheduling continuous perfusion
Switch medium: If desired, switch flow to reservoir containing medium 2.

Flow profiles can be steady, pulsatile or custom, e.g. mimic the cardiac rhythm
Results
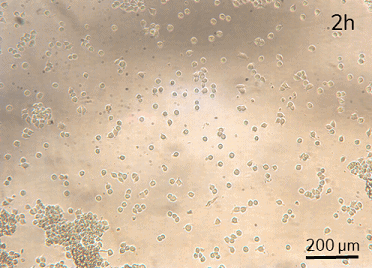
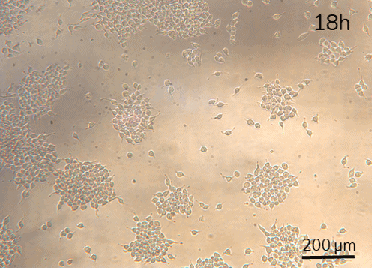
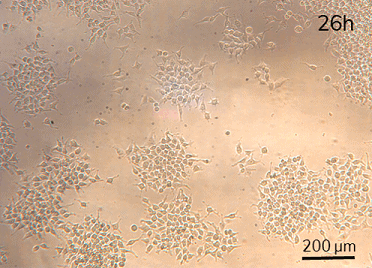
HeLa cell attachment and proliferation (t=2h, 18h, 26h). Perfusion (intermittent at 20 µL/min for 10 min per hour) was started at t=18h.
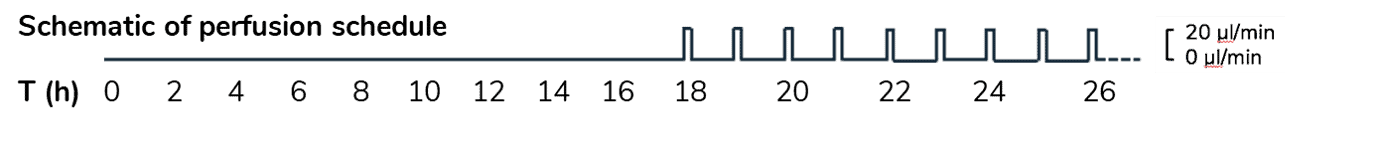
Hints & tips
| Our team of experts can help you perform your experiment, improve the configuration of the setup for your specific application and tackle any issues you could face. We can help you:
CONTACT US |
Application note written by Lisa MUIZNIEKS, Subia BANO, Camila BETTERELLI GIULIANO and Jessica AYACHE.
Acknowledgement: This work was done thanks to the funding of European Union’s Horizon 2020 research and innovation programme (PANBioRA project, grant agreement No 760921; MECH-LoC project, MSCA grant agreement No 793749; MTOAC project, MSCA grant agreement No 795754; Protomet project, MSCA grant agreement No 813873).

Microfluidics knowledge
Do you want tips on how to best set up your microfluidic experiment? Do you need inspiration or a different angle to take on your specific problem? Well, we probably have an application note just for you, feel free to check them out!









 Job
Job Collaborations
Collaborations Customer
Customer Other
Other
